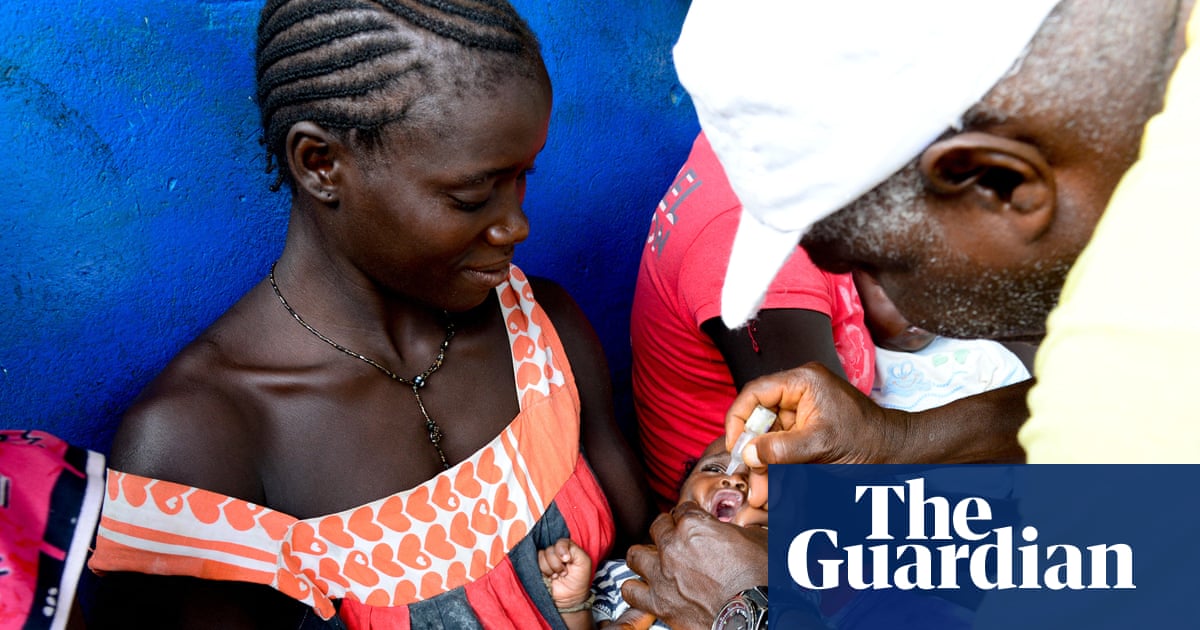
New details are leading experts to fear that an “unethical” vaccine trial in
Guinea-Bissau is the “prototype” for studies under
Robert F Kennedy Jr, secretary of the US department of health and human services (HHS) and longtime vaccine critic.At the center of US vaccine policy is an unlikely set of Danish researchers whose work on the health effects of vaccines has been called into question. The study in
Guinea-Bissau would have looked at the overall health effects of giving hepatitis B vaccines by only vaccinating half of the newborns in the study at birth despite an 18% prevalence rate in adults of the illness, which can lead to serious and sometimes fatal health consequences.
Stand Up for Science, a science and health nonprofit in the US, sent an investigator to
Guinea-Bissau to look at public records and interview experts. The organization met with members of Congress on 19 February to share these results in an unreleased report, obtained by the Guardian, that raises concerns about how deeply the
Bandim Health Project is enmeshed in public health in
Guinea-Bissau and the challenges to conducting ethical research in this setting – with immense repercussions for how US research will be carried out under Kennedy.“We are fearful that this is a prototype for other studies,” said
Colette Delawalla, founder of
Stand Up for Science. The US could fund global studies with the similar ethical concerns as the
Tuskegee experiment five or 10 or 100 times a year, she said. “It could be extraordinarily deadly.”
Stand Up for Science held nationwide rallies on Saturday to protest moves like these.Danish researchers behind the now-suspended hepatitis B vaccine trial have run Bandim in
Guinea-Bissau for 48 years, but they are now facing new questions about their previous work – most recently, a study published in Vaccine detailing several instances where the researchers appeared to conduct studies and then release partial results or none at all.The group has deep ties with the current US administration; Kennedy has cited
Peter Aaby, one of the researchers, as formative to some of his own views on vaccines.
Christine Stabell Benn, another of the researchers, was included on the hepatitis B working group of the advisory committee on immunization practices, which means she helped determine the evidence behind the committee’s decision to end the universal birth recommendation. Stabell Benn also hosted a podcast with Tracy Beth Høeg, a sports physician turned top official with the US Food and Drug Administration (FDA) who said she wants to end “unnecessary” vaccines.The researchers are “deeply, deeply connected in
Guinea-Bissau,” said Magda Robalo, former minister of health and president of the Institute for Global Health and Development in
Guinea-Bissau. “They are embedded in the system.”Bandim “is the government”, one source told the
Stand Up for Science investigator. The
Guinea-Bissau ethics committee is seen as “a friends club”, the investigator found.The ethics committee charges a fee for each study review, which is “problematic” because it could influence the members to approve the protocols, Robalo said. And the committee is not equipped to review research like this, she added.Informed consent is extremely challenging to obtain in
Guinea-Bissau because of low literacy rates and language barriers, Robalo said. The phrase in the local creole for hepatitis B and any other illness causing jaundice is “febri amarelu”, or “yellow fever” which is also the name of a separate disease. “How can there be informed consent if there isn’t even good language to describe to the people what they’re protecting themselves against by getting or not getting the vaccine?” Delawalla asked.“
Guinea-Bissau does not have a single credible institution in public health research at this point in time,” Robalo said. That also means many local researchers work with Bandim on studies, which creates potential conflicts of interest. For example, Armando Sifna, the current director of public health in
Guinea-Bissau, was affiliated with Bandim for more than a decade – as recently as December, when he was identified as working with Bandim and the national public health institute in
Guinea-Bissau simultaneously.That’s “very common”, Robalo said. Yet the local researchers have little power to influence what is studied and how, she said. While the Danish researchers argue that Bissau-Guineans want this trial to proceed, “there is tokenism here”, Robalo said. “Those people don’t hold the power for decision making.”Following the public outcry over the trial, the University of Southern
Denmark has paused “all work related to the study” while it is reviewed, said Ole Skøtt, dean of health sciences at the university. He said he has contacted the WHO’s research ethics review committee to conduct an independent ethical evaluation.The Guardian’s inquiries to minister of health Quinhin Nantote were not answered by press time. Stabell Benn requested questions by email but did not respond by press time.The US House committee on energy and commerce detailed concerns about the hepatitis B trial in a 6 February letter to Jim O’Neill, then the acting director of the US Centers for Disease Control and Prevention (CDC), calling the trial “ethically disturbing and scientifically unsound” and asking for the release of documents related to the CDC’s role in the study.The fact that CDC channeled federal funding to Kennedy’s associates without transparency or meaningful review “suggests that the agency’s grantmaking process may be seriously degraded in service of an anti-vaccine, ideological agenda”, the letter said.Tedros Adhanom Ghebreyesus, director-general of the World Health Organization (WHO), said at a news conference on 11 February that it was “unethical to proceed with this study”, and the organization released a statement on 13 February outlining its “significant concerns” over the study’s “scientific justification, ethical safeguards, and overall alignment with established principles for research involving human participants”.“It’s not going to happen, period,”
Guinea-Bissau’s foreign minister Joao Bernardo Vieira told Reuters on 18 February.Yap Boum II, a senior official at the Africa Center for Disease Control (CDC), told journalists on Thursday that Africa CDC is providing support to
Guinea-Bissau in reviewing the suspended trial – but the bigger issue should be rolling out birth doses of hepatitis B vaccine as soon as possible, and the discussion should “move to the comprehensive response”, Boum said. The evidence is clear, and recommending the vaccine to all babies at birth is “a high priority” for Africa CDC, said Landry Dongmo Tsague, director of primary health care at Africa CDC.Previously, Kennedy, citing Aaby’s work on vaccines in
Guinea-Bissau, yanked support for Gavi, the Vaccine Alliance, which was supporting the rollout planned for 2027. Nantote recently said the birth-dose campaign would be delayed until 2028. But African officials, including in
Guinea-Bissau, are exploring making the hepatitis B vaccine available to all newborns by 2027, Tsague said.The US allocated $1.6m to the hepatitis B trial, which had matching funding from the Pershing Square Foundation as well as the Bluebell Foundation. “They could actually just take this money and vaccinate every newborn for the next 10 years,” Delawalla said.“The study is paused for review,” an HHS spokesperson said. “CDC will continue to work with its partners to determine whether the study may be approved by the relevant home-country authorities.”The details of the trial have resonated across Africa. “Countries around the continent have been alerted to this,” Robalo said. They are working to improve their own research capabilities outside of the “asymmetric relationship between Global North research institutions and the Global South”, she said.