E(
European Medicines Agency (EMA)
OrganizationThe EMA is not directly mentioned in these articles, which focus on FDA approvals and Grifols' Biopharma IPO.
Mentions:1
7 Days:0
About
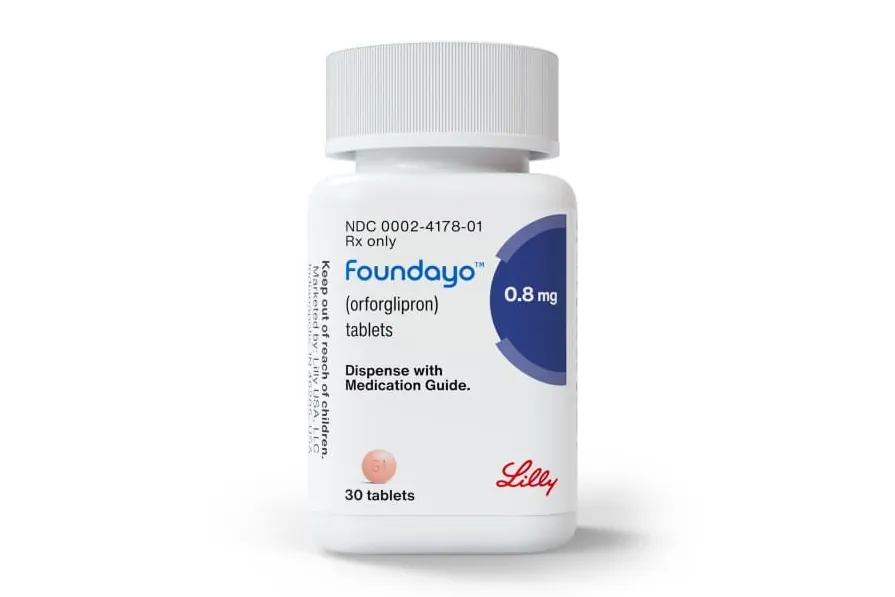
The European Medicines Agency (EMA) is not explicitly mentioned in the provided news articles. The articles primarily focus on the U.S. Food and Drug Administration (FDA) and the Spanish pharmaceutical company Grifols. One article details the FDA's approval of a new oral obesity medication from Eli Lilly, highlighting the competition in the obesity treatment market. The other articles discuss Grifols' plans to launch an Initial Public Offering (IPO) for its Biopharma business in the United States. While the EMA is a crucial regulatory body for pharmaceuticals in Europe, these specific news items are centered on developments within the U.S. market and the strategic decisions of a European company operating there. Therefore, the EMA's direct relevance to these articles is limited.
Last updated: May 7, 2026

